In a low-key laboratory in Baltimore, researchers are wagering that future pain relief could come from living nerve cells rather than tablets.
The premise is almost science fiction: produce human neurons from stem cells, shape them into miniature biological “sponges”, and place them into worn joints so they absorb pain signals before the brain registers them.
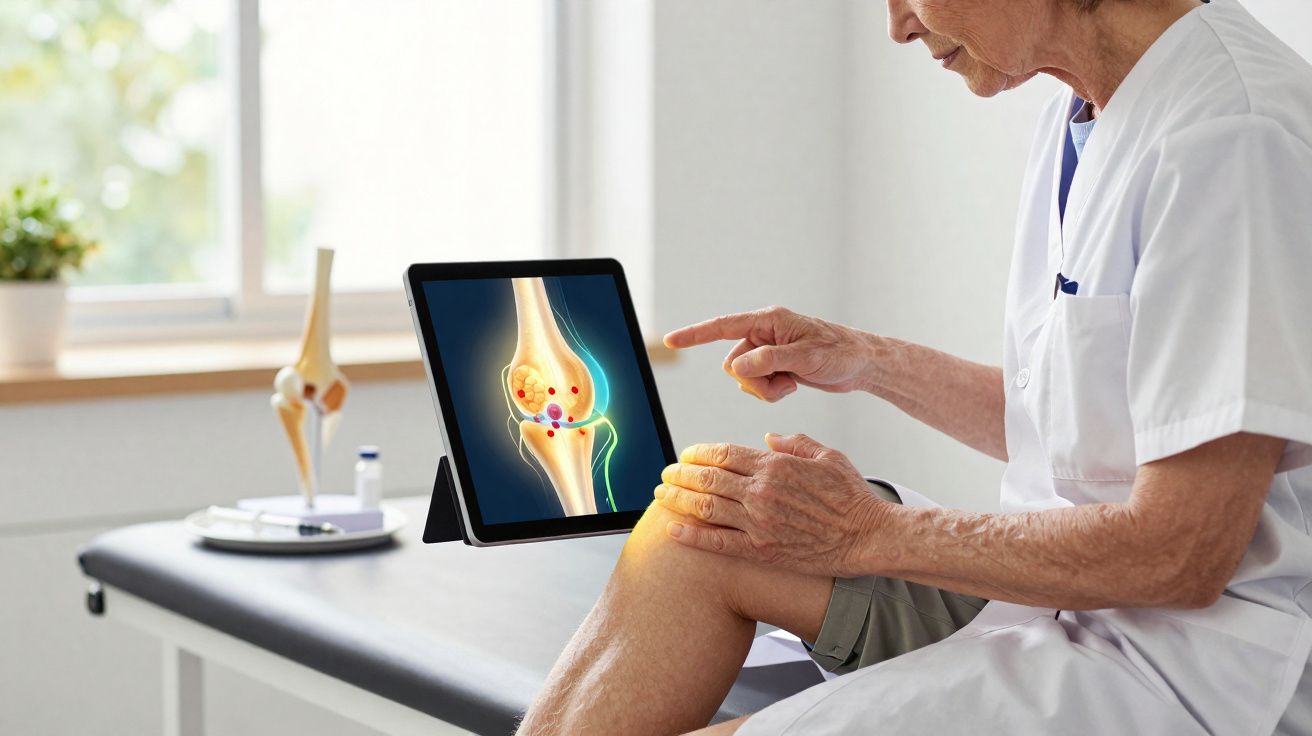
A new way to silence aching joints
A joint project between the Lieber Institute for Brain Development and Johns Hopkins University, alongside biotech start-up SereNeuro Therapeutics, has presented an experimental strategy for osteoarthritis pain that diverges strongly from today’s medicine-first approach.
Rather than saturating the whole body with anti-inflammatories or relying on opioids, the group creates specialist pain-detecting neurons from induced pluripotent stem cells (iPSCs). Those cells are then delivered directly into an arthritic joint, where they sit amid degraded cartilage and inflamed tissue.
"Rather than blocking the nervous system from above, these lab-grown neurons aim to calm pain right where it starts."
The findings, shared as a preprint on bioRxiv, remain preclinical and have so far been confined to animal studies. Even so, the idea is attracting interest because it appears to address chronic pain and joint deterioration together through a single, cell-based treatment.
How lab-grown neurons become “pain sponges” (SN101 osteoarthritis)
Induced pluripotent stem cells (iPSCs) are mature cells that have been reprogrammed back into a stem-cell-like state. From there, scientists can steer them towards almost any cell type. Here, the cells are directed into nociceptive sensory neurons-the same category that ordinarily detects painful stimuli.
SereNeuro’s engineered neurons, named SN101, are adjusted to act like natural human pain fibres. They display markers including CD200 and a range of receptors and ion channels-TRPV1, SCN9A, P2RX3-that are fundamental to pain signalling.
After implantation into an osteoarthritic joint in mice, the SN101 cells remain within the joint space and react to inflammatory molecules produced locally.
"The cells act less like a chemical blockade and more like a biological buffer, soaking up inflammatory cues before they trigger full-blown pain circuits."
Absorbing inflammatory signals instead of numbing nerves
During osteoarthritis, factors such as TNF‑α, IL‑1β and PGE2 surge within the joint. These substances stimulate nerve endings, sustain inflammation and help drive ongoing pain. The implanted neurons are intended to detect and bind to these mediators.
In laboratory assays, when SN101 neurons are exposed to these inflammatory drivers, they respond strongly and then lower the concentration of the factors in the surrounding liquid. The team characterises this as a “sponge-like” behaviour: the cells actively capture and dampen signalling molecules, rather than simply switching off nerve conduction.
This local buffering seems to translate in mouse studies. Behavioural assessments of movement and sensitivity suggest that animals given the neuronal implants show a marked drop in joint pain behaviours in roughly two weeks. The benefit persists for several months, with no sign of nerve destruction or complete loss of sensation.
- Site of action: inside the affected joint
- Main target: inflammatory mediators that activate pain-sensing nerves
- Goal: lessen pain while keeping normal nerve function intact
- Approach: durable cell therapy rather than repeated drug dosing
From pain relief to joint repair
What caught the researchers off guard was that the implanted neurons appear to do more than reduce pain-they also seem to encourage repair within the injured joint.
In osteoarthritic mice treated with SN101 cells, microscopic analysis showed thicker cartilage and improved quality of the bone just beneath the joint surface compared with untreated mice. Osteophytes (bony outgrowths) were also less prominent.
The team linked these changes to a mix of growth factors released by the implanted neurons, including BDNF (brain-derived neurotrophic factor), NGF (nerve growth factor) and VEGF (vascular endothelial growth factor).
"By releasing these growth signals over time, the neurons create a more nurturing environment for cartilage, bone and local nerves."
Common options such as corticosteroid injections can relieve pain for a time but, when used repeatedly, may speed up cartilage deterioration. By contrast, the SN101 approach is intended to provide steady pain reduction while shifting the joint microenvironment towards repair rather than further breakdown.
Why this matters for millions living with osteoarthritis
Osteoarthritis affects hundreds of millions of people globally and remains a major driver of disability. Typical care often follows a familiar sequence: lifestyle guidance, oral pain relief, stronger anti-inflammatories, steroid injections and, ultimately, joint replacement surgery.
Every stage involves compromises. Tablets act throughout the body rather than at the joint alone. Opioids carry risks of dependence. Surgery can be highly effective but is invasive, expensive and not appropriate for everyone. A precisely targeted, long-acting therapy delivered at joint level could change how clinicians approach long-term joint pain-especially for people considered too young or too frail for replacement surgery.
The roadblocks between mouse joints and human knees
The developers of SN101 are careful not to overstate progress. The programme is still preclinical, and moving a treatment this complex into human care raises significant scientific and regulatory hurdles.
A central issue is immune compatibility. If the cells are not made from a patient’s own tissue, the immune system may treat them as foreign and attack them. Keeping cells implanted over the long term also prompts concerns about uncontrolled growth or unforeseen effects on nearby nerves and tissues.
SereNeuro and its academic collaborators are now focused on standardising how the neurons are produced, assessed and stored. Batch-to-batch consistency is essential: identical markers, the same responsiveness to inflammatory molecules and an equivalent safety profile.
| Key challenge | Why it matters |
|---|---|
| Immune response | Rejection could wipe out the cells or cause dangerous inflammation. |
| Long-term safety | Engineered cells must not form tumours or damage healthy nerves. |
| Manufacturing quality | Each vial of cells must meet strict potency and purity criteria. |
| Regulatory pathway | Authorities will treat this as an advanced therapy, with heavy scrutiny. |
Initial human studies-expected to concentrate on severe knee osteoarthritis-are being prepared for the coming years. These first-in-human trials are expected to prioritise safety and tolerability, with pain and mobility measures serving as important secondary outcomes.
What “endoneural” really means in this context
The researchers describe their mechanism as provoking an “endoneural” response. In plain terms, that means the effect begins from within nerve tissue itself, rather than via an external drug that acts on a receptor or enzyme from the outside.
Put simply, the joint receives a small, built-in upgrade to its nervous system. The implanted neurons monitor the same chemical signals as native nerves, but they behave differently-buffering excess inflammatory messaging and releasing factors that soothe and support surrounding tissue.
What this could look like in the clinic
If later trials are successful, the patient pathway could look unlike today’s repeated injection cycle. Rather than attending every few months for another corticosteroid or hyaluronic acid injection, someone with advanced knee osteoarthritis might undergo a single procedure in which SN101 neurons are injected into the joint using imaging guidance.
Across the following weeks, pain could ease gradually as the cells establish themselves and begin absorbing inflammatory molecules. Mobility might improve not only because discomfort drops, but also because modest changes in cartilage and bone quality help stabilise the joint. The unresolved issue is durability-whether the benefit would last months, years, or require repeat implantation.
Risks, expectations and who might benefit first
As with any cell therapy, the risks are meaningful. Infection at the injection site, immune reactions, and unforeseen shifts in nerve sensitivity all require close surveillance. People taking potent immunosuppressive medicines, or those with a history of joint infection, may be excluded from early-stage trials.
Conversely, individuals with severe osteoarthritis who have exhausted standard options-or who are not candidates for joint replacement-may be among the first to consider this approach once safety is demonstrated. A living implant that both reduces pain and supports tissue health is likely to be particularly attractive to people caught between ineffective tablets and major surgery.
At present, SN101 is still an ambitious experiment. Even so, it points to a wider change in pain medicine: moving away from broad nervous-system suppression and towards precisely engineered, cell-based treatments that act where the problem actually sits-inside the joint itself.






Comments
No comments yet. Be the first to comment!
Leave a Comment